Acid Rain
causes
What is acid rain and how is it caused?
|
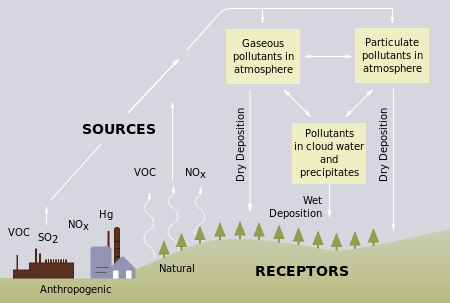
Acid Rain is caused by pollutants that make the rain acidic. Acidic Decomposition is a more accurate term used to describe Acid Rain for it's two parts, which can appear in many forms. Acidic Decomposition is in two parts which can both be transported by the wind; wet and dry (refer to diagram to bottom-left). Both forms are harmful to humans, animals and the environment. > Wet refers high acidity in the forms of rain, snow, fog or sleet. Because of it's forms, it flows on or through soil and affects the environment directly. Run off from the dry form adds to the acidity of streams and lakes that's water is then used for acid rain (precipitation). > Dry refers to high acidity in the form of gas or dust. Half the acidity of the dry forms falls back to earth from the atmosphere, and the particles can be washed away from surfaces and the run off from this contaminates streaks and lakes to become more acidic than the rain that came down. |
|
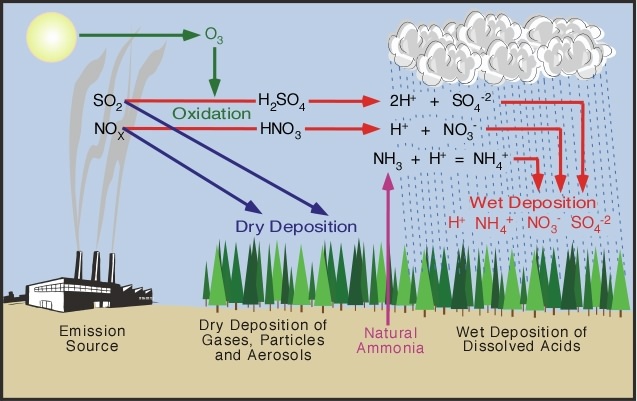
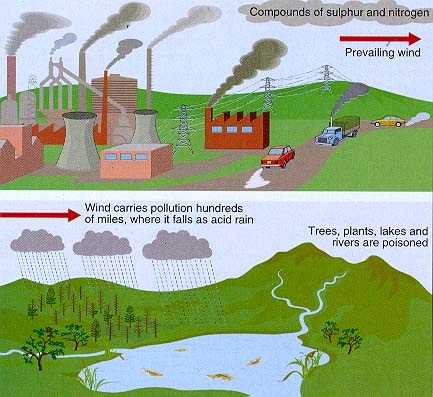
The predominant cause of Acid Rain is: Sulfur Dioxide (SO2) and Nitrogen Oxides (NOx). It is when these two gases react in the atmosphere with Water (H2O), Oxygen (O), and other chemicals react and form acidic compounds, Acid Rain occurs. A simple labelled diagram to the right, demonstrates how the cycle of pollutants swiftly reaching lakes and streams to poison the environment. |