acid rain
Damage
Ways damage by acid rain can be stopped or reduced
|
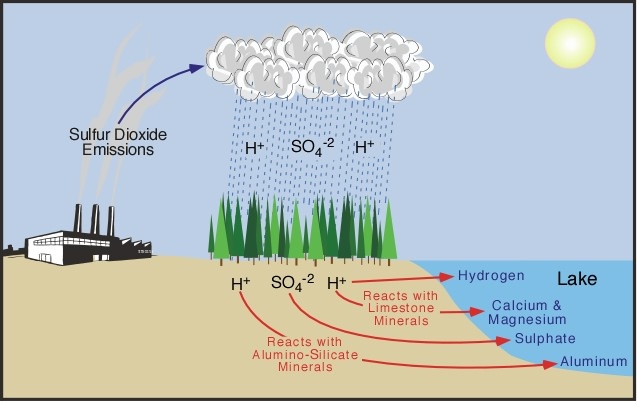
A more alternate method is to neutralise waters that have a low pH, high acidity, content to assist in the prevention of rain becoming acidic. When acid rain flows through soil that is very basic or high in alkaline properties, a reaction occurs to naturally neutralise the acid. *These soils contain limestone and carbon carbonate. If the acid rain is in a frozen form of wet composition then neutralisation can't occur. Other factors such as the type and thickness that the limestone and carbon carbonate are present in can determine whether or not the acid can be effectively neutrialised. This is because soils that don't weather consist of no or little bases, thus neurtalisation can't happen.
*Continuation of limestone and carbon carbonate neutralisation 1. Sulfuric acid and limestone react in a neutralization reaction. 2. Calcium sulfate is now soluble in water, therefore the limestone dissolves. 3. The Hydrogen ions from the original acid are converted into water. |
Acid Rain can only be reduced if there are less or no pollutants that cause rain to become acidic since there is no definite way of stopping acid rain. This includes using a lot less vehicles or machines that render air pollution, usually those that burn coal or fossil fuels.
Alternative methods can usually be carried out to deter the issue, such as using electric-fueled cars or analog vehicles (such as bicycles). If we are able to stop or reduce the amount of air pollution released into the atmosphere then there would gradually be less acid rain around the world. |